 Usability Engineering)”Company organisation chart””Job cards” and “Staff cards” according to ▶️ Procedure.Management of Normative and Regulatory RequirementsĮnsure that responsibilities and authorities are defined documented and communicated.ISO standards) Management of Normative and Regulatory Requirements according to ▶️ Procedure. 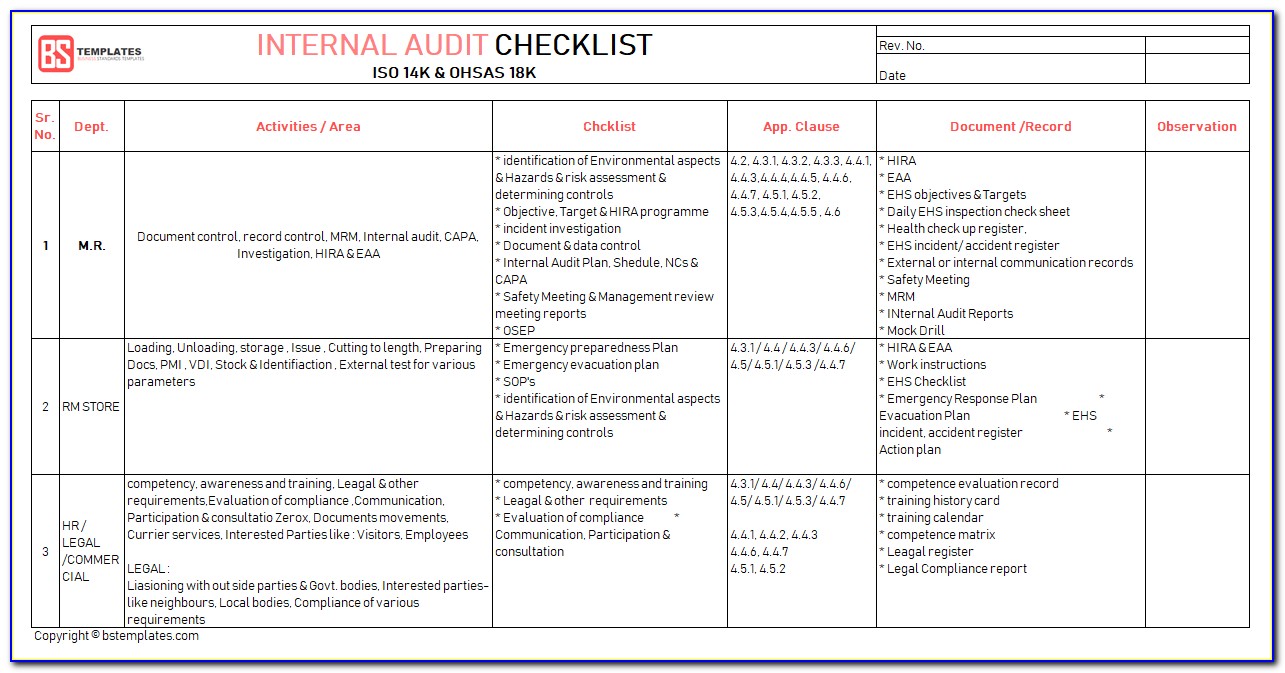
Management commitments and references to evidence of implementation are summarised below :Įnsure compliance with applicable regulatory and normative requirementsĭefinition of the tasks of the person responsible for ensuring compliance with regulations.List of regulations followed by the company (see □ Watch does not use ECM as part of its operationsĮxclusions are very rare, they must be justified by the implementation of another standard.Ĭontrol of monitoring and measuring equipment The working conditions and devices do not present a risk of contamination to personnel or devices Particular requirements for implantable medical devices The company does not use computer software used in production, or the company does not provide services 
The company does not offer Servicing activities The company does not offer installation activity Quality assurance does not cover design activitiesĭesign and development verification shall… Working conditions and devices do not present a contamination risk to personnel or devices The company does not use ECM in its operationsĮnvironmental working conditions are not critical to the safety of employees, the outcome of their activities or products conformity The following table identifies the requirements of ISO 13485:2016 that are not applicable to our company and justifies these non-applicabilities : ISO 13485:2016 requirements not applicable The QMS scope includes the activities of design, production, distribution, installation, maintenance, eliminationof (the reference or family of your devices). Take over the scope of your certification as defined with your Notified Body. Marketing of own-name MD with provision of instructions for use only in electronic format. Part of the supply chain of a MD from another manufacturer. Within the framework of the applicable regulatory requirements, the company assumes the role of : design, market, maintain) a (the type of device you offer).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
